Recombinant peptides are therapeutics produced from the recombinant DNA method, a technique which allows for much longer and more complex peptides to be produced. Production starts with fermentation using a microbial host, such as the bacterial microorganism E. coli, or a yeast-based microorganism such as S. cerevisiae or, more recently, the methylotrophic Pichia pastoris.
These microorganisms are engineered to express the recombinant peptide of interest during the fermentation process. Unlike the synthetic method of peptide production, the recombinant method produces a significant amount of host cell debris, glycosylated proteins, and other impurities that must be removed after harvest. In addition, recombinant peptides are prone to dimerization, aggregation, and fibrillation. Therefore, the recombinant production process requires multiple steps of downstream processing – including several chromatography steps – to achieve the desired purity. It is not unusual for an ion exchange chromatography (IEX) step plus one or two reversed phase chromatography (RPC) steps to be implemented in GMP production.
Silica gel is the most common substrate used for reversed-phase peptide purification due to its robust nature and proven resolving capability; however, not all silica gels are created equally.
Purification by Reversed-Phase Chromatography
Here, we will focus on the reversed-phase steps. There are several considerations when selecting the ideal stationary phase for recombinant peptide purification. Silica gel is the most common substrate
used for reversed-phase peptide purification due to its robust nature and proven resolving capability; however, not all silica gels are created equally. An ideal reversed-phase silica gel should contemplate
the following:
- Alkaline stability: since the crude material is loaded with many different impurities, the columns will necessarily require regular cleaning with caustic agents between cycles. CIP is a widely used procedure to remove self-aggregated, fibrillated peptides from the inlet side of the silica bed in the column. This regeneration procedure must be repeated periodically to maintain resolution, separation of peaks, and low column backpressure This emphasizes the need for a silica gel that is chemically compatible with alkaline conditions of up to 0.1M NaOH for cleaning.
- Binding capacity: this is a critical characteristic to improve the economics of the separation. With a higher loadability, a higher throughout of recombinant peptide can be purified in each cycle.
- Mechanical robustness: in some cases, the preparative or production-scale columns will need to
be unpacked and repacked to improve the column efficiency and prolong the media lifetime. This calls for a silica gel with high mechanical strength to withstand multiple axial compression packings.
A New Stationary Phase Designed for Complex Peptide Purification
Years of relentless research and development work at DAISOGEL led to the design and manufacturing of the most advanced silica-based stationary phase to solve these extraordinary challenges, which we call the PK Series.
With a 10 µm particle size for high resolution and a 100 Å pore size for enhanced loadability, we have also developed the PK Series with our advanced surface modification technology resulting in an unbeatable pH tolerance compared to other reversed-phase silica gel on the market. Further, its specially designed and manufactured silica backbone provides the extra mechanical strength to the silica particles. To test the capabilities of PK Series in real-life conditions, we performed a rigorous case study purifying a crude insulin sample.
Learn more in our new PK Brochure
CASE STUDY: Applying PK Series to Achieve >99% Purity on Crude Insulin
In this technical note we will demonstrate how the PK Series was able to bring a crude insulin feed of only 72.5% purity and raise the purity to over 99%. The initial purity of the crude sample was verified by an analytical HPLC analysis, as seen below.
As seen above, the low quality of the crude feed solution made the downstream modelling tricky. A well-executed polymer based strong cation exchange chromatography step (not shown) was used to bring the purity up to 88.0%. In order to achieve the final desired purity, two RPC steps using PK Series were employed.

RPC Step 1: ACN gradient against ammonium
sulfate / sulfuric acid on C8-PK Series
In the first RPC Step, a prep DAISOGEL SP-100-10-C8-PK column was overloaded with 15 g/L of IEX-purified insulin. With the target of 90% yield, an acetonitrile gradient was applied against a background of ammonium sulfate / sulfuric acid under the conditions shown below.
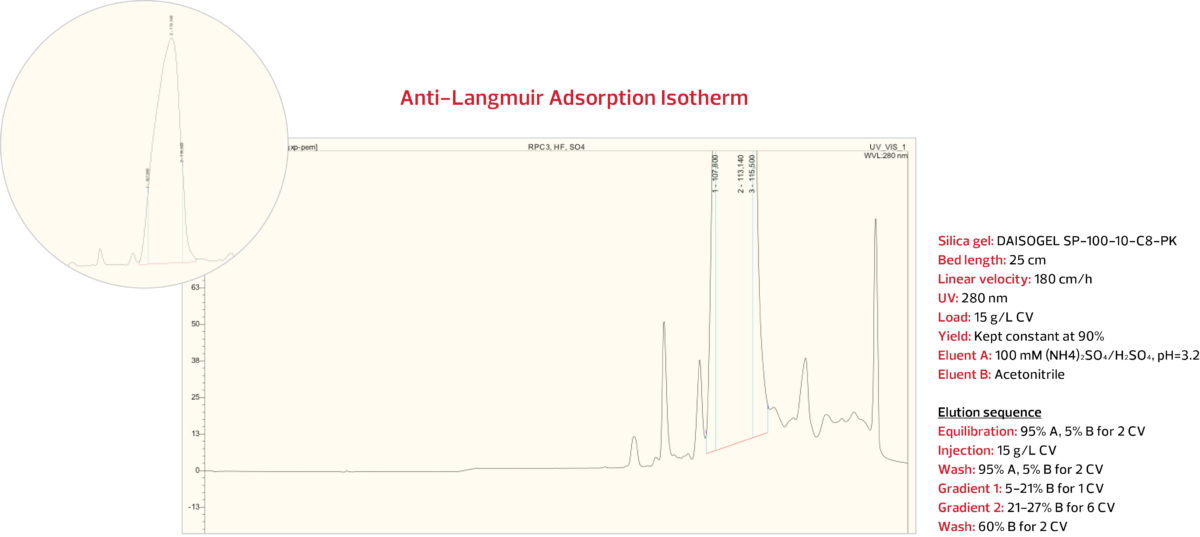
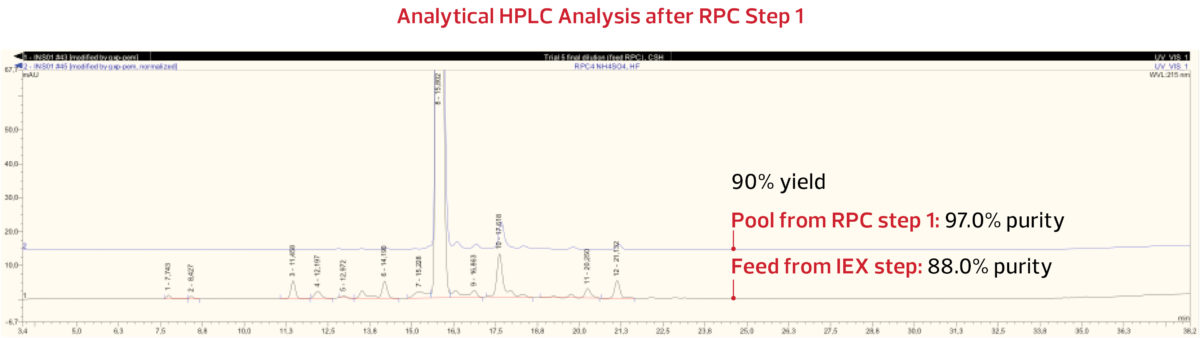
The insulin peak was collected and analyzed. The analytical HPLC results from the RPC Step 1 pool indicate
that with 90% yield, a purity of 97.0% was obtained, as shown below.
RPC Step 2: ACN gradient against ammonium
acetate / acetic acid on C8-PK Series
In order to achieve the desired >99.0% purity, a second RPC step was employed. A prep DAISOGEL SP-100-10-C8-PK column was overloaded with 15 g/L of RPC1-purified insulin. With the same target of 90% yield, an acetonitrile gradient was applied, but this time against a background of ammonium acetate / acetic acid, under the conditions shown below.
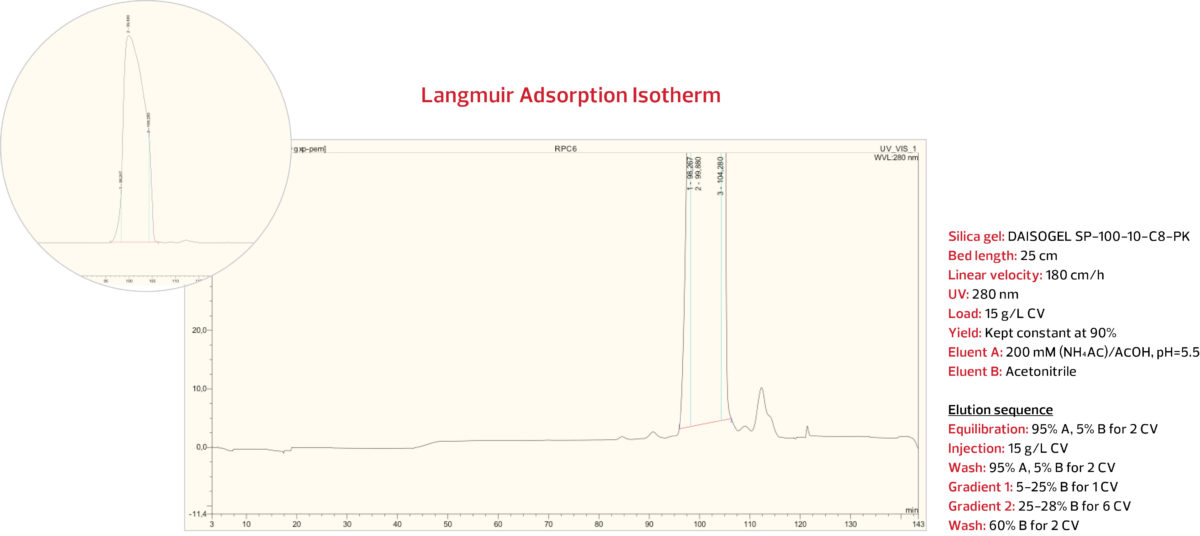
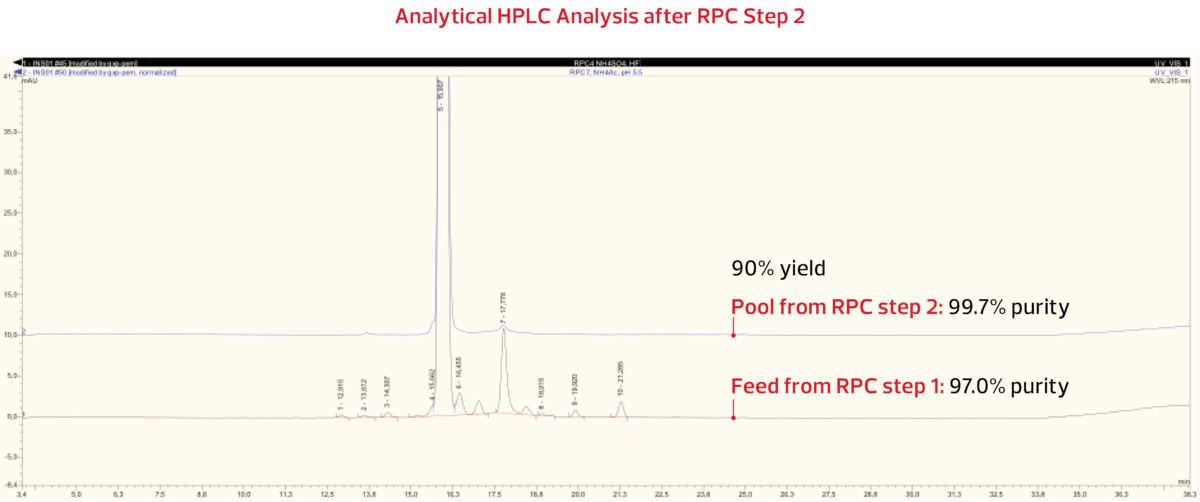
DAISOGEL SP-100-10-C8-PK
The insulin peak was again collected and analyzed. The analytical HPLC results from RPC Step 2 pool indicate that with 90% yield, a purity of 99.7% was successfully obtained, as shown below.
Conclusion
Recombinant peptides are complex by nature and challenging to purify, with insulin representing a classic example. Bringing the purity of such compounds to over 99% while not sacrificing yield is important for both product safety and economic reasons. DAISOGEL C8-PK Series silica gel was selected to purify a low-impurity crude insulin material due to its high surface area, loadability, mechanical strength, and resolving power.
By employing a two-step RPC purification after an IEX step, we were able to successfully achieve 99.7% purity while not sacrificing yield.
| Step | % of Purity |
|---|---|
| Crude feed | 72.5 |
| IEX | 88.0 |
| RPC1 | 97.0 |
| RPC2 | 99.7 |
